

Freeze-dried mixture of reverse transcriptase, RNase Inhibitor, the primer-probe sets of N gene and Human RNase P gene in the 0.2 mL 8-Strips qPCR tube.




【About SARS-CoV-2】
Coronavirus disease (COVID-19) is an infectious disease caused by the SARS-CoV-2 virus.It can be very contagious and spreads quickly. COVID-19 most often causes respiratory symptoms that can feel much like a cold, the flu, or pneumonia. COVID-19 may attack more than your lungs and respiratory system. Other parts of your body may also be affected by the disease. Most people infected with SARS-CoV-2 virus will experience mild to moderate respiratory illness and recover without requiring special treatment, but some people become severely ill.Some people including those with minor or no symptoms will develop Post-COVID Conditions – also called “Long COVID.” Influenza and COVID-19 are both contagious respiratory illnesses, but they are caused by different viruses.You cannot tell the difference between flu and COVID-19 by the symptoms alone because they have some of the same signs and symptoms. Nucleic acid tests are essential to distinguish between these diseases. Realtime PCR is an excellent, highly sensitive, specific and rapid technique for SARS-CoV-2 detection and is very useful for screening and confirmation of suspected cases under a wide range of circumstances.
【Test Principle】
The test tube is based on in vitro RT-qPCR combining fluorescent probing. The primer-probe sets of N gene was developed by China CDC and United States CDC[1-3]. All primer-probe sets were found to be highly specific for SARS-CoV-2, and no cross-reactivity with other respiratory viruses was observed[1-3]. The probes were attached by fluorophores at the 5’ end as reporter with VIC for N, N1 and N2 Gene, and quencher at 3’ end respectively. The test tube has internal control (Human RNase P gene[1-3]) with fluorophores ROX attached at 5’ end as reporter. During the PCR procedures, the DNA polymerase cleaves the probe at the 5’ end and separates the reporter dye from the quencher dye when the probes hybridize to the target DNA. This cleavage results in the fluorescent signal generated by the cleaved reporter dye, which is monitored real-time by the PCR detection system. Measuring the fluorescence intensities during Real Time PCR allows the qualitative detection of SARS-CoV-2 in specimens. The internal reference in the test tube is used to monitor the whole procedures including reagents and operation, to avoid false negative results.
【Application Data】
Samples: The sample is self-collected by four children with combined the oral and nasal swabs.
Reagents: TOROIVD® Self-Collection Kit(TPA-1611); TOROIVD® 5G Reaction Mix(TS-2500);
TOROIVD® SARS-CoV-2 Test Tube(TS-8001)
Tester: TOROUNIT® qPCR Cycler (TOROUNIT)
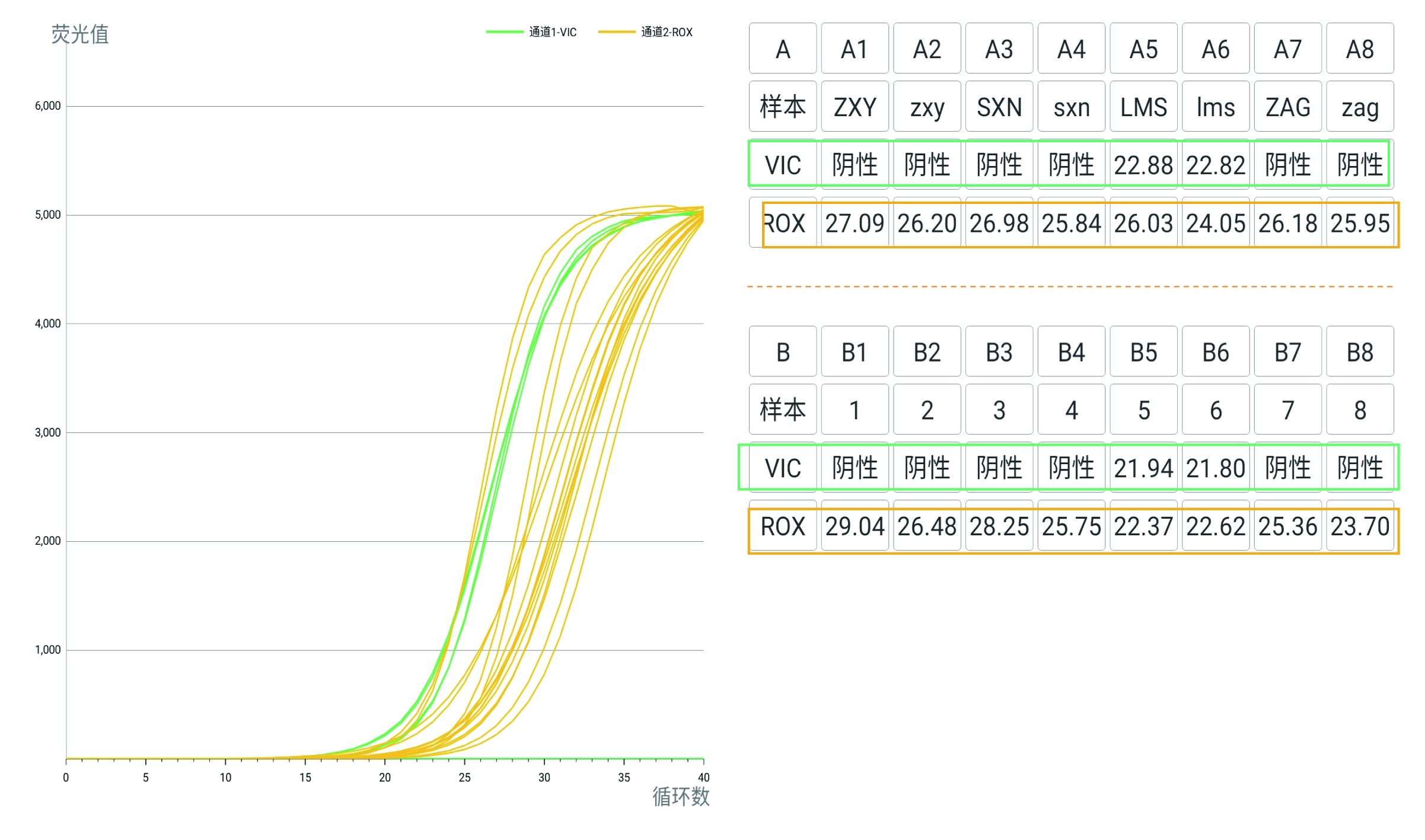
Yellow Curve-IC-ROX: RNaseP Gene monitored RT-qPCR assay
Green Curve-Target Gene-VIC: N Gene of SARS-CoV-2.
Results : A5/6&B5/6 are four repeats from LMS, LMS is positive of COVID-19.
A1-4, B1-4, A7/8&B7/8 are the tests from other three, They are negative of COVID-19.
【Storage】
Store the test tube at 2-8℃ in a dry environment for 24 months.
After opening, the remaining test tubes should still be sealed in a dry environment.
It is not recommended to use moisture absorbing test tube.
【References】
[1] Vogels CB, Brito AF, Wyllie AL,et al. Analytical sensitivity and efficiency comparisons of SARS-COV-2 qRT-PCR assays[J].Nature Microbiology[2023-08-22].
[2] Nalla AK , Casto A M , Huang MLW, et al. Comparative Performance of SARS-CoV-2 Detection Assays using Seven Different Primer/Probe Sets and One Assay Kit[J]. Journal of Clinical Microbiology,2020, 58(6).
[3] Jung, Y, Park, G.-S, Moon, JH, Ku, et al. Comparativeanalysis of primer-probe sets for RT-qPCR of COVID-19 causative virus (SARS-CoV-2)[J]. ACS Infectious Diseases,2020.
【Order infromation】
Cat NO. | Components | Size | Manual |
TS-8001 | 8 Tests/Bag |
For more details, please refer to TOROIVD Gene Test Tube Profile.
 | TOROIVD® XXX Test Tube.pdf |