

Freeze-dried mixture of reverse transcriptase, RNase Inhibitor, the primer-probe sets of B646L and IC gene, and IC DNA in the 0.2 mL 8-Strips qPCR tube.




【About ASFV】
African swine fever (ASF) is an infectious disease of domestic and wild pigs of all breeds and ages, caused by ASF virus (ASFV). The clinical syndromes vary from peracute, acute, subacute to chronic, depending on the virulence of the virus. Acute disease is characterised by high fever, haemorrhages in the reticuloendothelial system, and a high mortality rate. ASF cannot be differentiated from classical swine fever (CSF) by either clinical or post-mortem examination, and both diseases should be considered in the differential diagnosis of any acute febrile haemorrhagic syndrome of pigs. Bacterial septicaemias may also be confused with ASF and CSF. Nucleic acid tests are essential to distinguish between these diseases. Realtime PCR is an excellent, highly sensitive, specific and rapid technique for ASFV detection and is very useful for screening and confirmation of suspected cases under a wide range of circumstances.
【Test Principle】
The test tube is based on in vitro RT-qPCR combining fluorescent probing. The primer-probe sets of B646L gene was developed by WOAH [1], and were found to be highly specific for ASFV. The probes were attached by fluorphores at the 5’ end as reporter with VIC for B646LGene, and quencher at 3’ end respectively. The test tube has internal control DNA and primer-probe sets, and the probes with fluorophores ROX attached at 5’ end as reporter. During the RT-qPCR procedures, the DNA polymerase cleaves the probe at the 5’ end and separates the reporter dye from the quencher dye when the probes hybridize to the target DNA. This cleavage results in the fluorescent signal generated by the cleaved reporter dye, which is monitored by the real-time PCR detection system. Measuring the fluorescence intensities during Real Time PCR allows the qualitative detection of ASFV in specimens. The internal control is used to monitor the whole procedures including reagents, operation and qPCR cycler , to avoid false negative results.
【Application Data】
Samples: The sample is collected from a swine with a blood swab of tailhead.
Reagents: TOROIVD® Self-Collection Kit(TPA-1620); TOROIVD® 5G Reaction Mix(TS-2500);
TOROIVD® SARS-CoV-2 Test Tube(TS-8002R)
Tester: TOROUNIT® qPCR Cycler(TOROUNIT)
Results : The swine is the positive of ASFV.
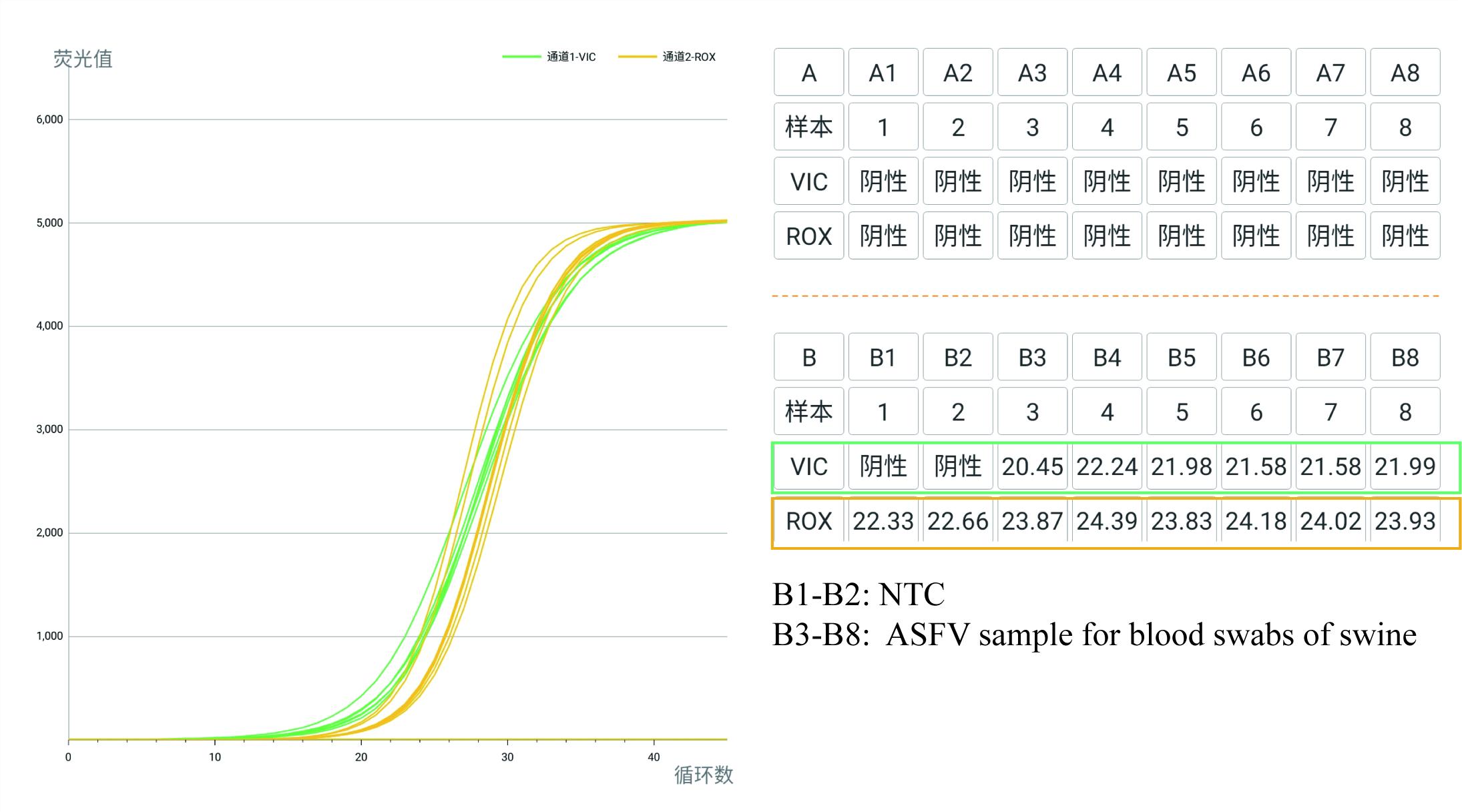
Yellow-Plot-ROX:IC monitored RT-qPCR assay
Green Plot-VIC:B646L Gene of ASFV
【Storage】
Store the test tube at 2-8℃ in a dry environment for 24 months. After opening, the remaining test tubes should still be sealed in a dry environment. It is not recommended to use moisture absorbing test tube.
【References】
[1] Manual of Diagnostic Tests and Vaccinesfor Terrestrial Animals, twelfth edition 2023, Chapter 3.9.1.World Organization for Animal Health, Updated 20/06/2023 .
【Order infromation】
Cat NO. | Components | Size | Manual |
TS-8002R | 8 Tests/Bag |
For more details, please refer to TOROIVD Gene Test Tube Profile.
 | TOROIVD® XXX Test Tube.pdf |